Clinical trial of ARCT-154 COVID-19 vaccine begins in Vietnam
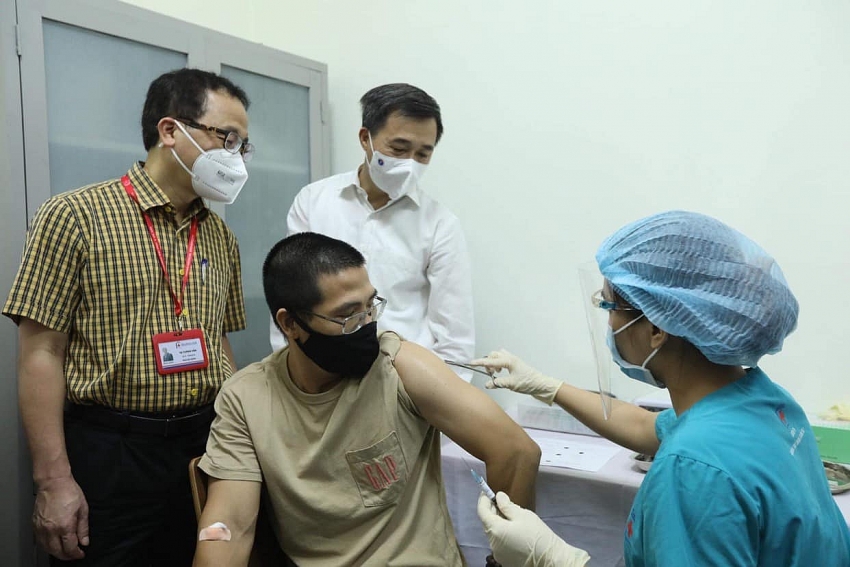 |
| Deputy Minister of Health Tran Van Thuan witnessed the administration of ARCT-154 vaccines for clinical testing on August 15 |
The first stage of clinical trials will be conducted on 100 volunteers aged 18 and older on August 15-16. The clinical trial will be carried out in three stages.
The second stage will be conducted on 300 volunteers from several provinces and the third stage will involve 20,600 volunteers.
Addressing the launch, Deputy Minister of Health Tran Van Thuan said, “I am delighted to witness the launch of clinical trials of the ARCT-154 vaccine using mRNA technology. I believe that the clinical trial will produce positive results, and Vietnam will have its own COVID-19 vaccines."
The COVID-19 vaccines using mRNA technology of Arcturus have been in clinical trials in the US, Singapore, and several other countries. ARCT-154 was found efficient against the highly infectious Delta variant and other known variants.
Earlier in August, the Ministry of Health approved the human clinical trial of the ARCT-154 vaccine. The vaccine technology was transferred to VinBioCare Biotechnology JSC, a unit of Vietnam's leading multisectoral conglomerate Vingroup, from Arcturus Therapeutics Holding Incorporation, US. This is a vaccine developed on mRNA technology to prevent COVID-19.
The ARCT-154 vaccine is the third COVID-19 vaccine to be produced in Vietnam. The two others are Nano Covax, and COVIVAC.
What the stars mean:
★ Poor ★ ★ Promising ★★★ Good ★★★★ Very good ★★★★★ Exceptional
Themes: Healthcare Platform
Related Contents
Latest News
More News
- Ministry of Health issues list of free regular health check-ups (May 13, 2026 | 18:34)
- Health-related community campaign launched in Hanoi (May 13, 2026 | 18:29)
- Health Ministry to implement free health check-ups (May 08, 2026 | 09:00)
- Revised Capital Law officially passed (April 24, 2026 | 10:20)
- Hanoi mulls time-based petrol motorbike ban on 11 streets (April 23, 2026 | 17:16)
- Government launches regular health checks alongside food safety crackdown (April 22, 2026 | 10:18)
- Vietnam recognised among top performers in World Bank human capital index (April 20, 2026 | 20:32)
- VCCI refutes US concerns over overcapacity and forced labour in Vietnam (April 17, 2026 | 17:22)
- Vietnamese among Asia’s most optimistic spenders (April 14, 2026 | 16:49)
- MoH urges hospitals to accelerate electronic medical records (April 13, 2026 | 14:19)



 Tag:
Tag:

























 Mobile Version
Mobile Version