Drugmakers build on early 2023 gains
Tran Tuc Ma, general director of Traphaco, the country’s second-largest publicly traded drugmaker, noted that it has received positive signals from the market so far this year.
 |
| Drugmakers build on early 2023 gains, illustration photo/ Le Toan |
“Last year marked a new development step for Traphaco as we began to implement business system restructuring, applying a new organisational model, and reforming sales and marketing, research and development, finance-planning, and other units,” he said.
Ma added that this year, Traphaco will focus on several big plans to tap into new opportunities. Specifically, the company will implement digital transformation in all company activities in a comprehensive manner, invest in upgrading production lines for its high-tech oriental medicine factory, carry out a feasibility assessment of GMP-EU standards in its Traphaco Hung Yen factory, and continue to implement its latest phase of tech transfer.
Traphaco is set to split off the entire sales system into the two groups of traditional and non-traditional medicines, invest in human and material resources, and separate sales policies with incentives to promote mutual development.
The company currently operates two factories in Vietnam – a smart western pharmaceutical factory and the first and largest GMP-WHO oriental medicine factory in the country, both in the northern province of Hung Yen.
Elsewhere, the nation’s fourth-biggest pharma firm Imexpharm saw its net revenue and pre-tax profit in January ascend 83.3 and 78 per cent on-year, respectively. The strong result is attributed to impressive growth in the over-the-counter channel and the ethical drugs channel, as well as good management of operating expenses.
In 2023, Imexpharm plans to accelerate digital transformation in its sales and marketing activities to increase efficiency and control possible risks; and strengthen cooperation with strategic partners to increase sales and expand the distribution network. The company will also focus on maintaining a prudent working capital management policy, minimising liquidity risks; shorten its operating days by reducing inventory and receivable days; and adjust and update policies to both attract and retain skilled workers.
Meanwhile, the top firm in Vietnam – DHG Pharmaceutical – is focusing on developing high-quality product lines that are qualified to replace foreign medicines while strengthening the company’s production capacity and increasing its competitiveness. Last year the group kicked off construction of its JAPAN/EU-GMP Betalactam factory, helping to increase its competitiveness.
Together with strategic and key products like Hapacol, Klamentin, Medlon, and Bocalex, the company has begun to distribute high-quality healthcare products from Japan.
Vietnam’s healthcare and pharmaceutical industry witnessed positive growth in 2022, driven by full operations at hospitals, thus increasing demands for drugs and tenders. Moreover, the government’s effort to make medical services more accessible at reasonable prices has also become a driving force for development.
With these conditions, the leading Vietnamese pharma giants made gains. Traphaco hit consolidated revenue of over $104 million in 2022, up from $94.35 million, while after-tax profit was $12.75 million, up 10.95 per cent on-year. This year, Traphaco plans to make revenues of $113 million and an after-tax profit of $14.17 million.
Imexpharm reported a rise in net revenues of 29.8 per cent on-year to $71.3 million, and its pre-tax profit increased by 26.3 per cent. This year, it is looking for pre-tax profit of $13.26 million, slightly up 1.1 per cent on-year.
Meanwhile, DHG made net revenues of $203 million last year, surpassing the year’s target by 11 per cent and rising by 17 per cent on-year. In 2023, the giant aims to fetch revenue of $217.4 million and a pre-tax profit of $49.13 million, up 3 per cent from 2022.
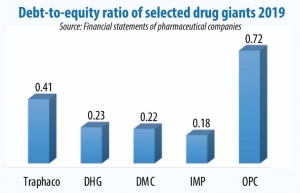
Amid continuing unpredictable changes worldwide, the pharmaceutical industry remains impacted here. Leaders said the major challenges come from a high reliance on imported raw materials, making up 80-90 per cent of total demand. Among the markets, imports from India and China make up 85 per cent of total imports. This means suffering changes in exchange rates, supply chain disruption, and import cost, thus increasing input costs by as much as 20-25 per cent.
Prescription drugs are expected to account for an increasing share of total pharmaceutical sales in the coming years, driven by rising medical treatment needs, the rollout of national health insurance, rising incomes, better healthcare infrastructure, and the development of generic drugs.
 | Drugmakers tackling disrupted supply chain While gaining advantage in the first quarter of 2020 amid the coronavirus outbreak, Vietnamese drug giants are predicted to face challenges in the upcoming months due to faults in the global ingredient supply chain. |
 | Drugmakers fight to attain approval Switzerland’s drug maker Novartis Pharma Services AG has announced a GMP qualification for Lek Pharmaceuticals d.d. within the scope of certification for medicines in Vietnam, but doubts have been raised about recognition in regards to the valuable and much-sought-after PIC/S-GMP and EU-GMP standards. |
 | Replaced regulation to hit global drugmakers International pharma giants like Pfizer, GSK, and Novartis are expected to be strongly hit soon by a price cut under Vietnam’s new tender rules for branded drugs, enabling local patients to receive an increase in pharmaceutical access at affordable prices. |
 | International investors scoop up major stakes in drugmakers Dealmaking and investment in pharmaceutical and healthcare businesses are increasing in scope as investors are vying for the controlling stake of key players. |
 | Drugmakers prepare to offer home vaccines to the world As Vietnam is stepping up its homegrown vaccine development, there is a high chance for Vietnamese vaccine producers to expand their presence in the global pharmaceuticals market. |
 | Drugmakers yearning for MA clarity Despite the government’s strong request for facilitation of marketing authorisation renewal, foreign drugmakers are still waiting for the renewal of expired authorisations, triggering concerns over a possible future shortfall of drugs. |
What the stars mean:
★ Poor ★ ★ Promising ★★★ Good ★★★★ Very good ★★★★★ Exceptional
Related Contents
Latest News
More News
- VCCI report highlights shift from control to facilitation in business regulations (June 03, 2026 | 08:00)
- Seaport operators deliver strong earnings despite global turbulence (June 02, 2026 | 19:13)
- Vietnam SuperPort® achieves highest facility security level under TAPA standards (June 02, 2026 | 18:46)
- Meey Global files confidential draft registration statement with SEC (June 02, 2026 | 16:00)
- Over 1,400 IP cases handled under PM's enforcement directive (June 02, 2026 | 12:26)
- China boosts imports of Vietnamese produce (June 02, 2026 | 07:00)
- AIG reinforces leading position through global expansion efforts (June 01, 2026 | 18:00)
- GStar Summit 2026 explores human-centric AI to boost productivity (May 30, 2026 | 15:32)
- Personalised lung cancer treatment: From genetic technology to robotic surgery (May 29, 2026 | 19:43)
- NS BlueScope launches 2026 Vietnam Steel Architecture Awards (May 29, 2026 | 16:04)

 Tag:
Tag:





















 Mobile Version
Mobile Version