Over 1.2 million doses of AstraZeneca’s COVID-19 vaccine arrive in Vietnam
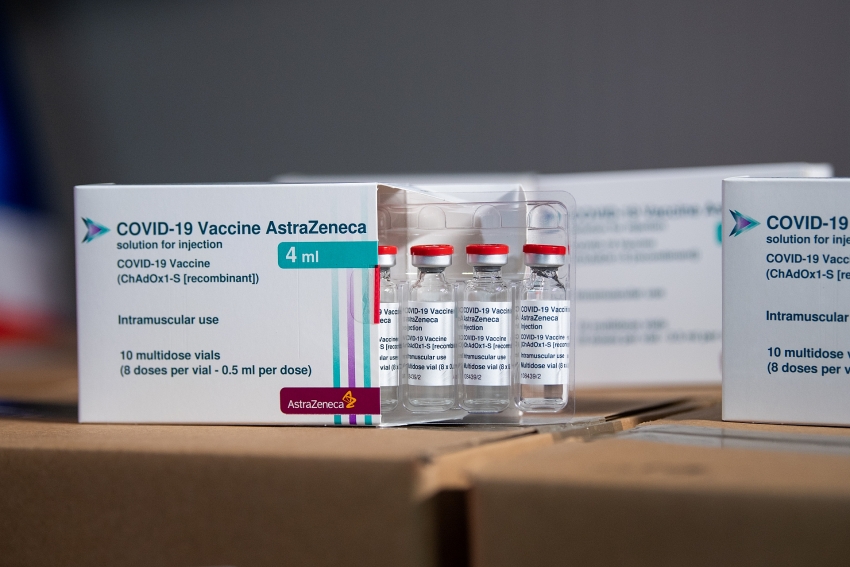 |
| Over 1.2 million doses of AstraZeneca’s COVID-19 vaccine arrived in Vietnam on August 19 |
To date, this agreement has brought over 6.7 million doses ofVaxzevria to Vietnam. Approximately 14.3 million doses of the vaccine have been delivered to the country through the APA, the COVAX Facility, and donations from other governments, accounting for nearly 62 per cent of the vaccine supply in the country.
In the afternoon of the same day, a call was also held between Vietnamese Prime Minister Pham Minh Chinh and the CEO of AstraZeneca, Pascal Soriot. Beyond vaccine access, the two sides also discussed the progress of AstraZeneca’s health system sustainability programmes in Vietnam, as part of the company’s efforts to address both infectious and non-communicable diseases and contribute holistically to the country.
Nitin Kapoor, chairman and general director of AstraZeneca Vietnam and Asia Area Frontier Markets said, “We were grateful to have the opportunity to speak with the PM today, following our meeting in late June. In response to his request for accelerated vaccine deliveries amidst this complicated COVID-19 outbreak, AstraZeneca has stepped up our supply to Vietnam since July to support the government’s national vaccination programme.”
 |
| Nitin Kapoor, chairman and general director of AstraZeneca Vietnam and Asia Area Frontier Markets, at a meeting with Vietnamese Prime Minister Pham Minh Chinh in June |
“We will continue to partner with the Ministry of Health, World Health Organization, UNICEF, and VNVC to deliver our vaccine to Vietnam as quickly as possible so that local communities can soon reunite with their families and return to their normal life,” he added.
To date, AstraZeneca has released more than one billion doses of its COVID-19 vaccine, developed in partnership with the University of Oxford, to more than 170 countries around the world at no profit.
AstraZeneca’s commitment to support broad and equitable vaccine access worldwide is reflected in its contributions to COVAX. The company was the first global pharmaceutical company to join the global initiative in June 2020, and has delivered more than 100 million doses of its COVID-19 vaccine through the COVAX Facility to date.
Since the first international launches in early 2021, the vaccine has helped to prevent hundreds of thousands of hospitalisations and saved tens of thousands of lives. The vaccine is generally well tolerated and around 80-90 per cent effective against severe disease and hospitalisation. The vaccine is highly effective against all variants of concern, including Beta and Delta.
AstraZeneca’s COVID-19 vaccine was the first to be authorised for and used in the Vietnamese government’s national vaccination programme. Globally, the vaccine has been granted a conditional marketing authorisation or emergency use in more than 80 countries across six continents.
What the stars mean:
★ Poor ★ ★ Promising ★★★ Good ★★★★ Very good ★★★★★ Exceptional
 Tag:
Tag:
Themes: Healthcare Platform
- Astrazeneca Vietnam eyes cooperation with MoH in new health financing solutions
- Advancing early prevention of paediatric respiratory diseases
- Ministry of Health issues list of free regular health check-ups
- Health-related community campaign launched in Hanoi
- Promomed eyes big steps in Vietnam’s pharma market
Related Contents
Latest News
More News
- Ministry of Finance and ADB discuss development cooperation priorities (June 03, 2026 | 18:25)
- Building trusted supply chains: Industry experts highlight security, connectivity and digitalisation (June 03, 2026 | 14:58)
- Vietnam accelerates capital market reforms to boost foreign investor access (June 03, 2026 | 11:58)
- Hong Hac City sets benchmark for integrated urban living (June 03, 2026 | 08:00)
- Vietnam SuperPort® achieves highest facility security level under TAPA standards (June 02, 2026 | 18:46)
- Vietnam and Philippines strengthen cooperation in digital technology (June 02, 2026 | 18:34)
- Meey Global files confidential draft registration statement with SEC (June 02, 2026 | 16:00)
- Over 1,400 IP cases handled under PM's enforcement directive (June 02, 2026 | 12:26)
- Aboitiz eyes deeper investment in Vietnam's energy, food security (June 02, 2026 | 08:00)
- China boosts imports of Vietnamese produce (June 02, 2026 | 07:00)


























 Mobile Version
Mobile Version