UK starts Moderna jabs as AstraZeneca probed
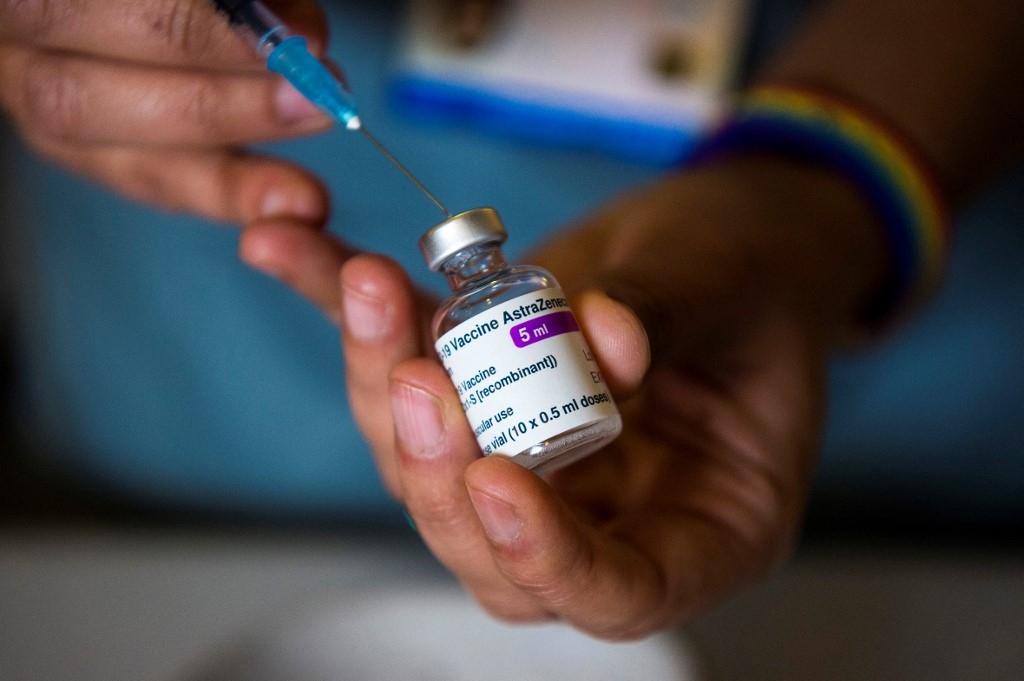 |
| In this file photo taken on January 07, 2021 Dr Nikki Kanani prepares to administer the Oxford/AstraZeneca COVID-19 vaccine, at the Sunrise Care Home in Sidcup, south east London. A British trial of the AstraZeneca coronavirus vaccine on children has been paused while regulators assess its possible link to blood clots, Oxford University, which helped develop the jab, said on April 6, 2021.(KIRSTY O'CONNOR / POOL / AFP) |
The Moderna vaccine, which is already being delivered in Europe and the United States, joined ones from AstraZeneca-Oxford University and Pfizer-BioNTech in Britain's armoury against Covid-19.
The first jabs of the two-stage Moderna inoculation were injected at a hospital in Wales, in a timely diversification of Britain's rollout that was hailed by Prime Minister Boris Johnson.
"We have ordered 17 million doses that will be going into arms across the UK in the coming weeks. Please get your jab as soon as you are contacted," he tweeted.
Supply problems for AstraZeneca had threatened to complicate Britain's inoculation drive this month, and concerns are building over a potential link between the jab and rare blood clots among a small number of recipients.
Oxford University said late Tuesday that it had paused a British trial of the AstraZeneca vaccine on children.
The university said the trial had posed "no safety concerns", but that it was awaiting more data from Britain's Medicines and Healthcare products Regulatory Agency (MHRA) before restarting the study.
The MHRA says it is looking into the cases of clotting, amid reports that the regulator may join some countries in the European Union in restricting access in younger age groups.
The MHRA reported over the weekend that there had been 30 blood clotting cases, seven fatal, out of 18 million doses administered in Britain.
The European Medicines Agency (EMA) is also looking anew at the issue, but so far the World Health Organization insists the jab is safe.
Kent Woods, a British former head of both the MHRA and EMA, told LBC radio that the risks of Covid were much higher and he had "no reservations" about the AstraZeneca vaccine.
But Maggie Wearmouth, a member of the government's Joint Committee on Vaccination and Immunisation, told the Daily Telegraph that "perhaps slowing things down" with the rollout "until we're absolutely certain" might be wise.
Any delays could imperil the British government's phased easing of its current coronavirus lockdown, with all adults due to receive a first vaccine dose by the end of July.
What the stars mean:
★ Poor ★ ★ Promising ★★★ Good ★★★★ Very good ★★★★★ Exceptional
 Tag:
Tag:
Related Contents
Latest News
More News
- International media spotlight Vietnam’s National Assembly election (March 16, 2026 | 09:41)
- Citi report finds global trade transformed by tariffs and AI (February 25, 2026 | 10:49)
- ASEAN unveils digital outlook, AI readiness research (February 11, 2026 | 11:52)
- Vietnam and Laos step up urban and infrastructure cooperation (February 04, 2026 | 16:04)
- Space Summit 2026 opens in Singapore amid alignment calls (February 02, 2026 | 17:00)
- PM inspects APEC 2027 project progress in An Giang province (January 29, 2026 | 09:00)
- Philippines plans higher rice imports to stabilise supply (January 28, 2026 | 11:11)
- France supports Vietnam’s growing role in international arena: French Ambassador (January 25, 2026 | 10:11)
- Foreign leaders extend congratulations to Party General Secretary To Lam (January 25, 2026 | 10:01)
- Russian President congratulates Vietnamese Party leader during phone talks (January 25, 2026 | 09:58)





















 Mobile Version
Mobile Version