Vietnam approves sixth COVID-19 vaccine for emergency use
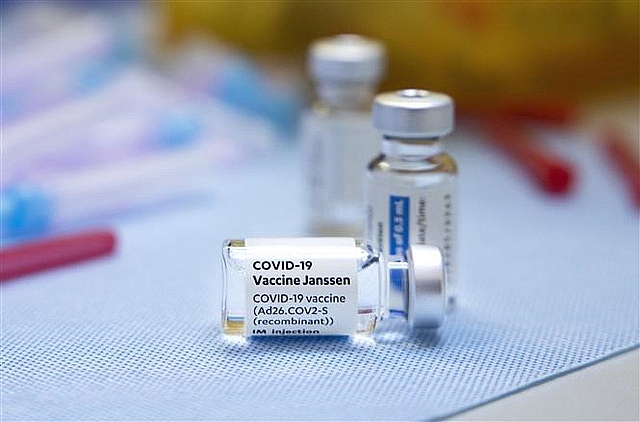 |
| The COVID-19 Vaccine Janssen |
The vaccine is produced by Janssen Pharmaceutica NV (Belgium) and Janssen Biologics BV (the Netherlands).
This is the sixth kind of COVID-19 vaccine that Vietnam has approved with conditions for emergency use. The five others are Comirnaty of Pfizer, A2D1222 of AstraZeneca, Sputnik-V of Gamalaya, Vero-Cell of Sinopharm, and Spikevax of Moderna.
The move helps the country increase the vaccine supply amid worst outbreak in the southern region so far.
The MoH has assigned the Drug Administration of Vietnam (DAV) to grant the import licence for the vaccine when it receives the documents of authorised importers in line with the prevailing rules.
The ministry also asked the Agency of Science, Technology and Training to select qualified units to assess the efficacy and safety of the vaccine.
Meanwhile, the National Institute of Vaccine and Medical Biology will need to verify the factory certificate of Janssen vaccines.
What the stars mean:
★ Poor ★ ★ Promising ★★★ Good ★★★★ Very good ★★★★★ Exceptional
Themes: Healthcare Platform
Related Contents
Latest News
More News
- Ministry of Health issues list of free regular health check-ups (May 13, 2026 | 18:34)
- Health-related community campaign launched in Hanoi (May 13, 2026 | 18:29)
- Health Ministry to implement free health check-ups (May 08, 2026 | 09:00)
- Vietnam recognised among top performers in World Bank human capital index (April 20, 2026 | 20:32)
- VCCI refutes US concerns over overcapacity and forced labour in Vietnam (April 17, 2026 | 17:22)
- Vietnamese among Asia’s most optimistic spenders (April 14, 2026 | 16:49)
- MoH urges hospitals to accelerate electronic medical records (April 13, 2026 | 14:19)
- Hanoi considers easing pickup truck restrictions to support businesses (April 02, 2026 | 15:52)
- Female leadership to steer Vietnam's future (April 02, 2026 | 14:56)
- Hanoi targets rabies elimination as pet meat trade revenues plummet (March 25, 2026 | 10:31)



 Tag:
Tag:


























 Mobile Version
Mobile Version